 Front Bioeng Biotechnol ; 2020 Nov 23
Front Bioeng Biotechnol ; 2020 Nov 23
角膜位于眼睛的最外层,很容易受伤。眼部化学烧伤占眼部损伤的11.5-22.1%,碱烧伤是化学烧伤中最常见的。羊膜(AM)是胎盘的最内层,在怀孕期间保护胎儿。目前,AM产品因其独特的生物学特性而被美国食品和药物管理局批准用于帮助眼表重建。由于其出色的生物学性能,AM移植是治疗角膜碱烧伤的有效药物。AM作为一种临时覆盖材料,可以促进角膜上皮化,抑制炎症。AM的生物学特性归功于羊膜上皮细胞的各种生长因子。然而,在AM准备过程和长期储存后,生长因子将逐渐丢失和不足,无法保持活性。
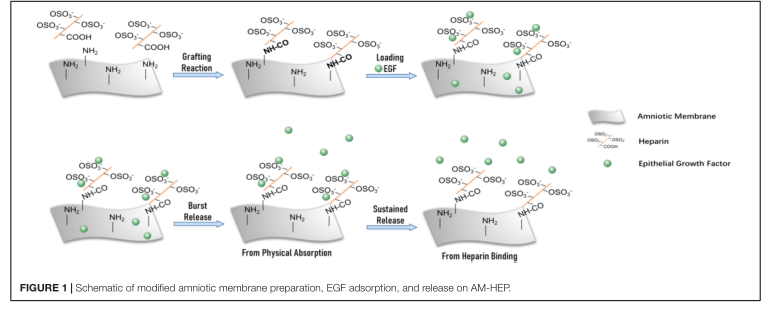
中山大学眼科中心袁进教授团队致力于角膜及眼表的研究,提出一种改良的羊膜(AM),即肝素改性的羊膜AM(AM-HEP),用于通过表面移植肝素补充和缓释生长因子,用于治疗眼化学烧伤。对AM-HEP的肝素接枝率和稳定性、微观结构、物理性质和表皮生长因子(EGF)的缓释进行了表征。评价了生物相容性和促进角膜上皮细胞生长和迁移的能力,并与商品化的生物羊膜进行了比较。在小鼠角膜碱烧伤模型上,观察了AM-HEP@EGF联合EGF对小鼠角膜碱烧伤的治疗作用。结果表明,在AM中引入了肝素,并在37℃下保持了3周以上的稳定性。与未改性的AM相比,AM-HEP的改性过程并未影响其微观结构和物理性能。EGF与AM-HEP结合迅速有效,缓释时间可达14天以上。AM-HEP@EGF能明显促进角膜上皮细胞的生长和迁移,与未修饰的AM和对照组相比,差异有统计学意义(P<0.05)。体内移植AM-HEP@EGF的角膜上皮化速度明显快于未经处理的对照组。与对照组相比,AM-HEP@EGF组的角膜炎症程度较轻,透明程度较高。体内外实验结果表明,AM-HEP@EGF能显著提高治疗效果。研究结果表明:AM-HEP@EGF通过促进角膜上皮伤口愈合,在角膜碱烧伤中具有潜在的临床应用价值。
Cornea is located on the outermost layer of the eye and is easily injured. Ocular chemical burns are responsible for 11.5–22.1% of ocular injuries. Alkali burn is the most common among chemical burns and considered as ophthalmic emergencies. Amniotic membrane (AM) is the innermost layer of the placenta and protects a fetus during pregnancy. Currently, AM products were approved by the Food and Drug Administrationthat are clinically used to help the ocular surface reconstruction due to its unique biological characteristics. AM transplantation is an effective medical therapy for corneal alkali burn due to its outstanding biological performances. As a temporary covering material, AM can promote corneal epithelialization and inhibit inflammation. The biological performances of AM are due to various growth factors from amniotic epithelial cells. However, growth factors will be gradually lost and insufficient and cannot maintain the activity after AM preparation process and long-time storage. The advantage of fresh AM is the growth factors is abundant, so it has more significant effects in anti-inflammatory, inhibiting scar formation, inhibiting neovascularization, and promoting epithelial cell proliferation.
Professor Yuan Jin and his group from Sun yat-sen university ophthalmology center presented a modified AM (AMHEP) for the supplement and sustained release of growth factor by surface grafting heparin for treatment of ocular chemical burns. Heparin grafting rate and stability, microstructure, physical property, and sustained release of epithelial growth factor (EGF) of AM-HEP were characterized. Biocompatibility and ability to promote corneal epithelial cell growth and migration were evaluated and compared with a biological amnion, which is available on the market in vitro. The therapeutic effects of AM-HEP combined with EGF (AM-HEP@EGF) in vivo had been evaluated in a model of mouse corneal alkali burn. The results indicated that heparin was introduced into AM and maintain stability over 3 weeks at 37◦C. The modification process of AM-HEP did not affect microstructure and physical property after comparing with non-modified AM. EGF could be combined quickly and effectively with AM-HEP; the sustained release could last for more than 14 days. AM-HEP@EGF could significantly promote corneal epithelial cell growth and migration, compared with non-modified AM and control group. Faster corneal epithelialization was observed with the transplantation of AM-HEP@EGF in vivo, compared with the untreated control group. The corneas in the AM-HEP@EGF group have less inflammation and were more transparent than those in the control group. The results from in vitro and in vivo experiments demonstrated that AM-HEP@EGF could significantly enhance the therapeutic effects. Taken together, AM-HEP@EGF is exhibited to be a potent clinical application in corneal alkali burns through accelerating corneal epithelial wound healing.
DOI: 10.3389/fbioe.2020.599800
王丽